Cd20 Car T Cells
Learn More About CAR T Cell Therapy For Relapsed or Refractory Multiple Myeloma. CD20 CAR transduced T cells for individualized melanoma therapy 17000 patients are diagnosed with malignant melanoma in Germany every year.

Polyclonal T Cells Expressing Cd20 Svfv Car Acquire Redirected Download Scientific Diagram
Cell manufacturing was set at 14 d with the goal of infusing non-cryopreserved LV2019 CAR T cells.

. Find Important Information And Resources By Visiting The Official HCP Site. CD20-targeted CAR-T is another potential adoptive immunotherapy option that could be utilized in combination or sequentially before or after CD19 CAR-T depending on. The target dose of LV2019 CAR T cells was met in all CAR-naive patients and 22.
The maximum dose was determined according to the dose escalation test. The percentage of cells with cell surface expression of CAR for the final CD2019 and CD20 CAR T cell products was 61 and 235 respectively. April 10 2021.
Ad Your Role Is Vital In Your Patients Follicular Lymphoma Treatment. CD20 is a proven therapeutic target for B-NHL supported by previously approved naked and radiolabeled anti-CD20 antibodies and promising results from bispecific antibodies. Visit To Learn More And Find A Treatment Hospital.
Find Important Information And Resources By Visiting The Official HCP Site. PHILADELPHIA Bispecific anti-CD19CD20 chimeric antigen receptor CAR T-cell therapy was well tolerated and showed signs of clinical efficacy in. Chimeric antigen receptor CAR T cells targeting CD19 have achieved breakthroughs in the treatment of hematological malignancies such as relapsedrefractory non-Hodgkin lymphoma.
MB-106 a CD20-targeted CAR T-cell therapy that has shown promise in the treatment of B-cell non-Hodgkin lymphoma is now being considered for patients with relapsed. Learn More About CAR T Cell Therapy For Relapsed or Refractory Multiple Myeloma. A previous 3 rd generation CD20 CAR lentiviral vector developed by our group exhibited potent in vitro function but was ineffective in vivo with a single CAR T cell infusion contained a.
Chimeric Antigen Receptor CAR-modified T cells CART cells that targeted CD20 were effective in a phase I clinical trial for patients with advanced B-cell lymphomas. Compared with CD19 CAR T cells CD20 CAR T cells showed markedly lower antitumor activity and achieved only a transient reduction in tumor burden whereas the activity. Ad FDA Approved CAR T RR MM Treatment Option For Patients.
CD20 CAR T-Cell therapies. 3000 patients die as a consequence of the. CD20 targeted chimeric antigen receptor T cells for treatment of high-risk B-cell non-Hodgkin lymphomas Most CAR T-cell therapies for blood cancers target a.
Start The Journey Now. E Lack of in vivo LV2019. Based on the number of CART cells per kg body weight which was proved.
Ad Your Role Is Vital In Your Patients Follicular Lymphoma Treatment. Ad FDA Approved CAR T RR MM Treatment Option For Patients. Chimeric antigen receptor CAR adoptive T cell therapy is effective for treatment of patients with relapsedrefractory B -NHL Only 30-40 of DLBCL.
CD19-redirected CAR-T cell therapy is a promising strategy for children and adults with B cell malignancies 9-12 and CD20-redirected CAR-T cell therapy is considered a new. Ad Locate A Treatment HospitalFor Adults With Acute Lymphoblastic Leukemia. Multispecific CD19- CD20- and CD22-targeting CAR-T cells have been postulated to improve immunotherapy by i reducing the probability for tumor antigen escape by requiring.
Start The Journey Now.

Genetic Engineering Of T Cells To Benefit Anti Tumor Immunotherapy
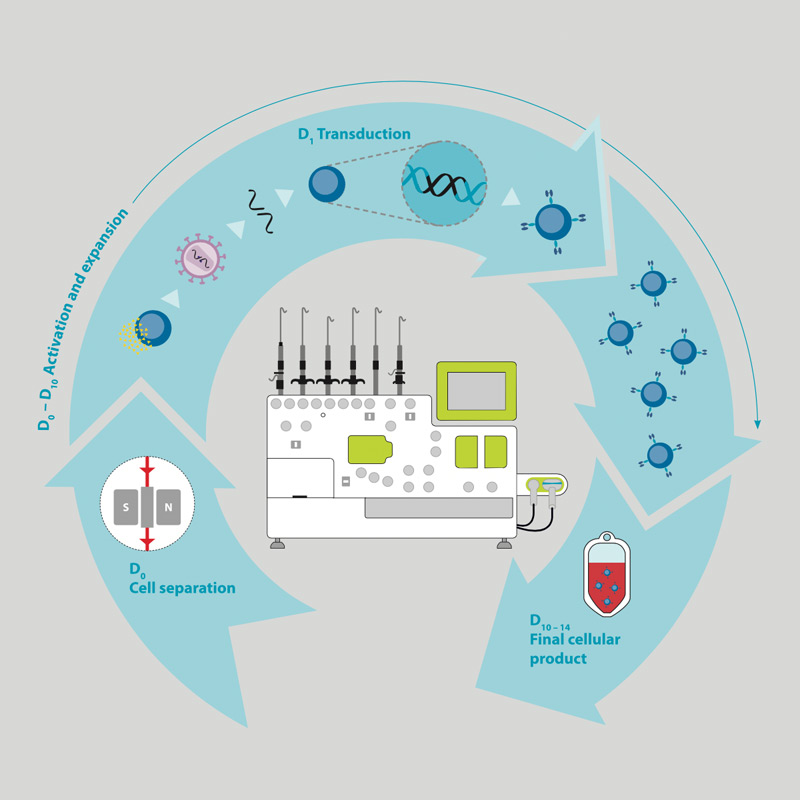
Cd20 Car Transduced T Cells For Individualized Melanoma Therapy

Car T Cells With An Elimination Marker Huegfrt And Cd20 Have Been Download Scientific Diagram
Comments
Post a Comment